The "core lifeline" of biopharmaceuticals—do you really understand the core value of the special gas supply system?
In the biopharmaceutical industry, there is a system that appears "invisible" but permeates the entire production process, determines the fate of the product, and relates to corporate compliance – it is the special gas supply system. Unlike the intuitive equipment such as laboratory petri dishes and workshop fermentation tanks, the special gas supply system is the "core hub" that supports biopharmaceutical production. Its design, performance, and operational stability directly determine the efficiency of pharmaceutical production, product quality, and market competitiveness, making it an irreplaceable key infrastructure in the biopharmaceutical industry. The so-called special gas supply system is a dedicated system specifically used for transporting, regulating, and purifying special gases (referred to as "special gases").
Its core function is to deliver various special gases needed in biopharmaceutical production (such as oxygen, carbon dioxide, nitrogen, etc.) with industry-standard purity, stable pressure, and flow rate, accurately, safely, and cleanly to every production node. It is not a simple "gas transport pipeline," but a comprehensive system that integrates purification, filtration, monitoring, regulation, and safety protection, serving as an "invisible but indispensable" core support in biopharmaceutical production.

A qualified special gas supply system must meet the stringent requirements of the biopharmaceutical industry for purity, stability, and safety. Its core components revolve around the five key functions of "purification, delivery, control, monitoring, and protection," with each element directly affecting the overall performance of the system and ultimately determining the production results.
Core Components
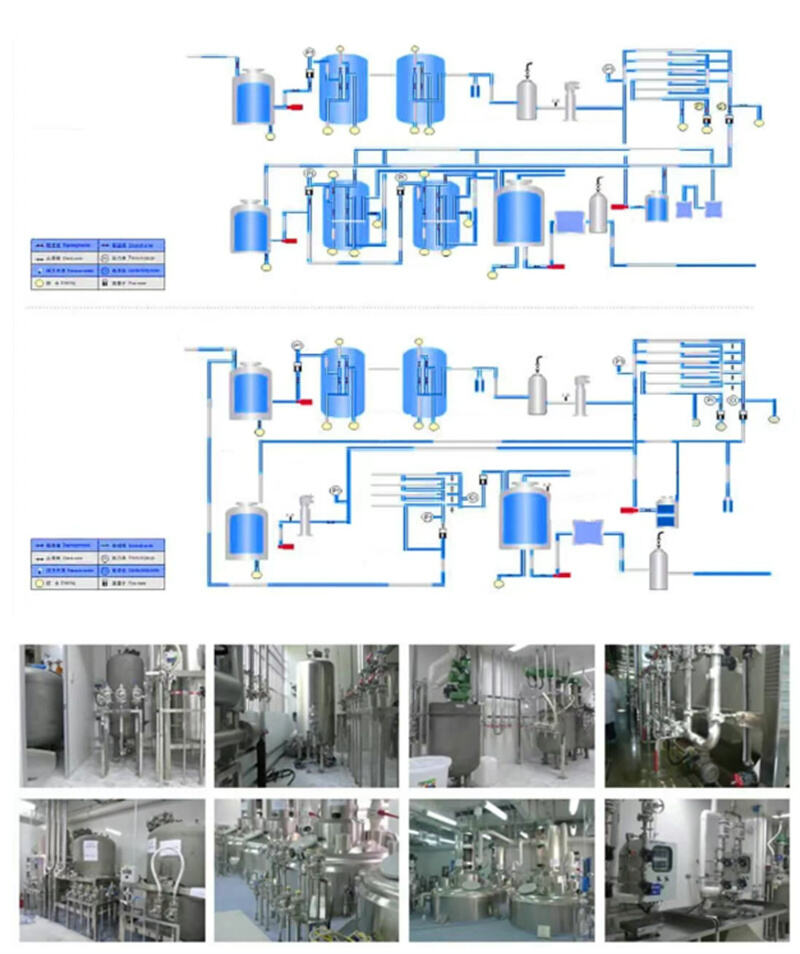
The core components of the special gas supply system can be divided into five major modules, which work together to ensure the accuracy and safety of gas supply: first, the gas storage module, which is used to properly store various special gas cylinders to prevent gas leakage and mixing risks; second, the purification and filtration module, which is the core of ensuring gas purity, using multi-stage filtration and purification processes to remove moisture, particles, and harmful impurities from the gas, ensuring that the gas purity meets standards; third, the transportation pipeline module, which uses 316L stainless steel and EP-grade electro-polishing technology, with an inner wall roughness as low as Ra≤0.2μm, reducing gas adsorption and impurity residue to prevent secondary contamination; fourth, the control module, which uses precise valves, flow meters, and pressure regulators to achieve stable control of gas flow and pressure, eliminating fluctuations; and fifth, the monitoring and protection module, which integrates real-time monitoring of pressure, flow, and temperature parameters, along with AI fault warning and explosion-proof, leak-proof design, to ensure system safety and data traceability.
Core Function
The core value of the special gas supply system lies in its three key functions, which safeguard biopharmaceutical production, making it distinct from ordinary gas systems: First, the precision purification function: addressing the "microgram-level" purity requirements of biopharmaceuticals, the system can increase the purity of special gases to over 9N (99.9999999%), a two-order-of-magnitude improvement over traditional gas systems, eliminating impurities in the gas that can cause product contamination from the source, ensuring product purity and efficacy; Second, the stable control function: it can provide continuous gas supply 24/7, precisely controlling the range of gas flow and pressure fluctuations, avoiding issues such as abnormal cell growth and production interruptions caused by unstable gas supply; Third, the safety and compliance function: strictly adhering to GMP (Good Manufacturing Practice) requirements, it achieves real-time recording of gas monitoring data and early warning of faults, ensuring the traceability of the production process, helping pharmaceutical companies pass compliance audits and avoid operational risks.
The performance of the special gas supply system directly determines its core value
A qualified special gas supply system must meet the stringent requirements of the biopharmaceutical industry for purity, stability, and safety. Its core components revolve around the five key functions of "purification, delivery, control, monitoring, and protection," with each element directly affecting the overall performance of the system and ultimately determining the production results.
Purity control capability: The "first line of defense" that determines drug safety
One of the core performance characteristics of a special gas supply system is its ability to control purity. In biopharmaceuticals, even impurities at the ppb level (one part in a billion) can lead to a decrease in drug purity, damage to efficacy, or even cause safety incidents. High-quality special gas systems, through professional purification modules and pipeline design, can effectively remove various impurities from the gas and maintain high purity stably; whereas inferior systems cannot achieve precise purification, which can not only result in the entire batch of drugs being scrapped but also expose pharmaceutical companies to compliance penalties.
Stable operational capability: The key to reducing production costs
Stable operation is a core requirement of the special gas supply system and a key factor in controlling production costs for pharmaceutical companies. A high-quality system can provide long-term, uninterrupted, and stable gas supply with minimal pressure and flow fluctuations, effectively reducing production interruptions and cell culture failures caused by system failures. Additionally, its gas loss rate can be reduced to less than 3%, a significant decrease compared to traditional systems (12% loss rate), thereby reducing special gas waste. Conversely, inferior systems frequently experience leaks and failures, which not only increase gas consumption and maintenance costs but also cause significant economic losses due to production interruptions.
Compliance Adaptation Capability: The "Hard Support" for Pharmaceutical Companies' Survival
The compliance adaptation capability of the special gas supply system directly affects whether a pharmaceutical company can pass GMP audits and achieve large-scale production. A high-quality system is designed strictly according to GMP requirements, integrating comprehensive monitoring and data recording functions to ensure gas purity and operational parameters are traceable, while also featuring comprehensive safety protection designs that meet industry regulatory requirements. A system that does not meet the standards will prevent the pharmaceutical company from passing compliance audits, losing its production qualifications, and ultimately being eliminated from the market.
 EN
EN
 AR
AR
 HR
HR
 CS
CS
 NL
NL
 FR
FR
 DE
DE
 IT
IT
 JA
JA
 KO
KO
 NO
NO
 PL
PL
 PT
PT
 RO
RO
 RU
RU
 ES
ES
 SV
SV
 TL
TL
 ID
ID
 VI
VI
 MT
MT
 TH
TH
 TR
TR
 AF
AF
 MS
MS
 AZ
AZ